Patients with heart failure with improved ejection fraction benefit from the SGLT2 inhibitor dapagliflozin: Study
With modern therapies for heart failure (HF) with reduced ejection fraction (HFrEF), some patients can improve their cardiac function during treatment. But despite this improvement in the ability of their hearts to pump, these patients with so called heart failure with improved ejection fraction (HFimpEF) remain at high risk for adverse outcomes.
Unfortunately, they have been excluded from virtually all clinical trials in heart failure and there has been little evidence about how best to improve clinical management for this growing patient population.
Researchers from Brigham and Women’s Hospital, a founding member of Mass General Brigham, and collaborators from the University of Minnesota and University of Glasgow have conducted an analysis that suggests that this patient population may further benefit from initiation of the SGLT2 inhibitor dapagliflozin, a heart failure medication that has received attention after presentations earlier this year on data from the randomized, controlled DELIVER clinical trial.
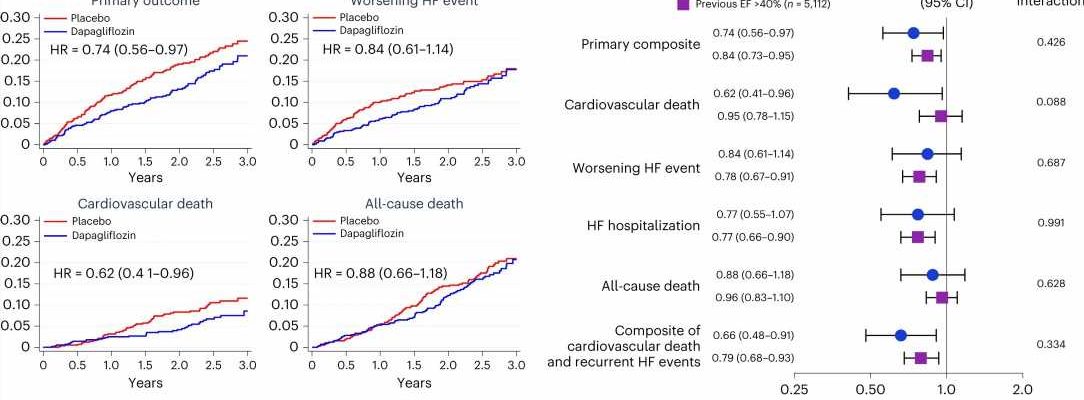
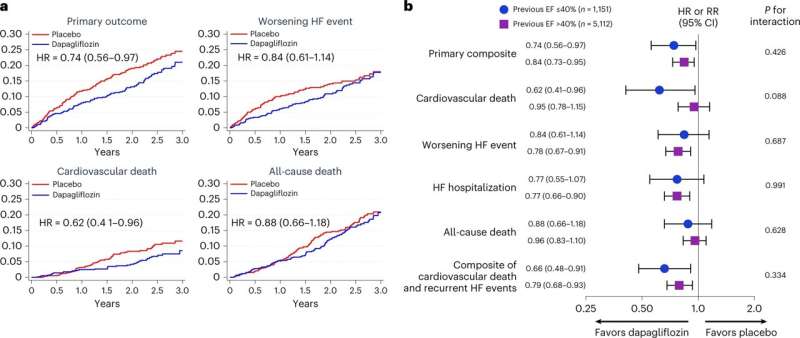
In a prespecified analysis of data from the DELIVER trial, researchers looked at outcomes for 1,151 patients with HFimpEF and found that dapagliflozin reduced the primary composite outcome, first worsening heart failure events, cardiovascular death and total worsening heart failure events.
“These are essentially the first large-scale randomized outcomes data in patients with heart failure and improved ejection fraction,” said co-corresponding author Scott D. Solomon, MD, of the BWH Division of Cardiovascular Medicine.
“As current therapy of heart failure with reduced ejection fraction gets better, and more and more patients show improvement, this group is becoming larger. These data suggest that addition of an SGLT2 inhibitor can benefit these patients and should inform treatment decision-making.”
The research is published in the journal Nature Medicine.
More information:
Orly Vardeny, Dapagliflozin in heart failure with improved ejection fraction: a prespecified analysis of the DELIVER trial, Nature Medicine (2022). DOI: 10.1038/s41591-022-02102-9
Journal information:
Nature Medicine
Source: Read Full Article