Inhibiting a certain enzyme in cryptococcus may halt infection spread
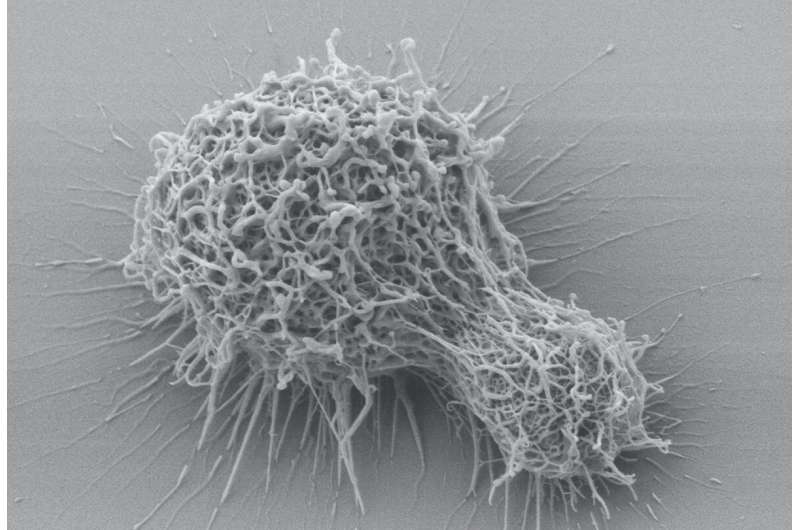
The last drug designed to more effectively treat dangerous systemic fungal infections, most often affecting immunocompromised patients, was developed over 20 years ago. Now an interdisciplinary team of investigators from Stony Brook University believe they have identified what may be a new approach toward developing another class of better antifungal agents. By inhibiting an enzyme called sterylglucosidase 1 (Sgl1) in a model of Cryptococcosis, the researchers found infection did not spread. They believe this enzyme can be a target for a new class of antifungal drugs. The research findings are published in Nature Communications.
Current antifungals have many drawbacks for patients including high toxicity and many drug interactions, so the need for new antifungals remains high. Cryptococcus neoformans is a fungal pathogen that, upon entering the lung and moving into the bloodstream, causes life-threatening meningoencephalitis in susceptible patients and is life-threatening.
The COVID-19 pandemic has placed a sharp lens on the dangers of infections that spread, particularly to the lungs. Cancer patients and other immunocompromised individuals with chronic conditions are much more susceptible to systemic fungal infections such as Cryptococcosis.
“Essentially by identifying selective small molecule inhibitors of Sgl1 in an experimental model, we found that the infection failed to spread to the brain and began to be cleared from the lungs within two weeks,” summarizes Mike Airola, Ph.D., Senior Author and Assistant Professor in the Department of Biochemistry and Cell Biology in the Renaissance School of Medicine at Stony Brook University.
“We believe based on our experimentation that by targeting Sgl1, we can create a new class of antifungal agents against this and possibly other fungal pathogens in order to prevent both primary and secondary infections,” says Nivea Pereira de Sa, Ph.D., First Author of the manuscript and post-doctoral fellow in the laboratory of Dr. Del Poeta. “We are taking the next steps to test this approach.”
Airola says that the research team involves a collaboration of scientists at Stony Brook through the Institute of Chemical Biology and Drug Discovery. The team includes microbiologists, cell biologists, immunologists and chemists. Additionally, the groundwork to this finding was led by Co-Senior author and SUNY Distinguished Professor Maurizio Del Poeta, MD, an internationally recognized expert in fungal disease. His lab demonstrated that Sgl1 is a fungal specific protein, and when mutated, the fungus is non-pathogenic.
The research team presented co-crystal structures of Sgl1 with two inhibitors. By combining traditional medicinal chemistry with computational approaches, they were able to unravel how these anti-Sgl1 compounds interact with the enzyme.
Source: Read Full Article


